
As a child, I always wanted a microscope.
I would have collected slimy waters from the scum ponds and murky puddles near my house. I would have brought them home and exposed them to the light of my microscope. I would then have peered deep into a secret world, where shady characters and alien forms lurked and traded.
It would be many years, when I was in college, before I finally witnessed this world—so alien, it might have inspired the science fiction books I wrote later as an adult. As it turned out, I was led to pursue a Masters of Science degree, studying periphyton (microscopic aquatic communities attached and associated with surfaces like rocks and plants) in local streams in the Eastern Townships of Quebec.
While my work focused on how diatoms (glass-walled algae) colonized surfaces, micro-invertebrates kept vying for my attention. Water fleas (cladocerans), copepods, rotifers, seed shrimps (ostracods) and water bears sang across my field of vision. They flitted, lumbered, wheeled and meandered their way like tourists lost in Paris. But this wasn’t Paris; I’d taken the blue pill and entered the rabbit hole…
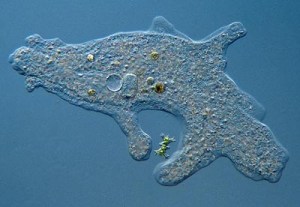
Aquatic habitats are home to strange and extraordinary tiny life forms that can best be seen through a microscope. Single-celled organisms include bacteria (Monerans), protozoans and algae. Scientists have estimated that over 50,000 different species of protozoans exist. The Paramecium, which most of us studied at school, is a Ciliate; its hundreds of cilia beat in unison to propel it through the water. Mastigorphorans, like Euglena and the colonial Volvox, use a whip like extension called a flagellum that rotates or sprials like a gondola’s sculling oar to propel the individual or colony. The protoplasm of the Amoeba (a Sarcodine) flows freely, forming pseudopods that creep forward and surround prey.
Aquatic habitats are also home to micro-invertebrates, multicellular animals that can barely be seen with the naked eye. Many average from 0.5 to 1 mm in size and resemble little white blobs; however, a scholar can distinguish each invertebrate by its unique movement.
Who’s Who in the Micro-Invertebrate World
Gastrotrichs are benthic invertabrates, commonly referred to as “hairybacks”. The short worm-like gastrotrich Chaetonotus lurks in the detritus of periphyton using adhesive glands to anchor itself and feeds on smaller creatures by sucking on its powerful pharynx. It glides and bends its long torso using cilia in slow, deliberate movements, looking for food among the organic debris. Freshwater gastrotrichs are parthenogenetic, producing unfertilized eggs that hatch and mature with a lifespan of a few days.
Copepods (meaning “oar-feet”) are tiny crustaceans with a tear-drop shaped body and long antennae. They live in many aquatic habitats from wet forests, damp moss, ephemeral ponds and puddles to the ocean depths. A copepod has a hard outer shell with a black or red eye-spot in the middle of its head. Copepods “jerk” through the water, often en masse as they feed on phytoplankton or prey on smaller invertebrates (like rotifers). Harpacticoid copepods eat organic detritus or bacteria. During adverse conditions, calanoid copepods produce dormant eggs and some can enter dormancy as nauplii (baby copepods or larvae) or copepodids (juveniles).
Ostracods, also called shrimp seeds, are cosmopolitan crustaceans with an oval hinged bivalve shell (carapace) beneath which their legs swim. Ostracods glide like underwater dirigibles in wet leaf litter, springs, ground water, puddles, small ponds and rivers, feeding on decaying organic matter and algae. Freshwater ostracods are mostly parthenogenetic (self-cloning). Hydra and small fish feed on them. Blue Sand ostracods glow blue at night, using a light organ that produces luminescent chemicals (either as predation defense or for mating).
Cladocerans, also called water fleas, resemble underwater birds with a single compound eye and six appendages attached to the thorax beneath a flattened transparent carapace. The cladoceran’s flamboyant antennae continually beat to keep the animal suspended and transport it in vertical “hops”—resembling a goldfinch in flight—as it filter-feeds on algae, protozoa, bacteria and decaying organic material. Cladocerans live in many types of aquatic habitats, ranging from acidic swamps to freshwater lakes, ponds, streams and rivers. Hydra and fish feast on this invertebrate.
Most of the time, cladocerans are all female, creating new female clones. You can see the eggs inside their transparent bodies. When conditions deteriorate (e.g. winter or a pond drying up) the females produce males. They mate, producing resistant dormant eggs, which will only hatch when better conditions arise.

Rotifers are cosmopolitan detrivores (they eat detritus) and contribute to the decomposition of organic matter. Rotifers create a vortex with ciliated tufts on their heads that resemble spinning wheels, sweeping food into their mouths. They often anchor to larger debris while they feed or inch, worm-like, along substrates. Some are sessile, living inside tubes or gelatinous holdfasts and may even be colonial. Rotifers reproduce by parthenogenesis (in the absence of mates), producing clones (like cladocerans). Resting eggs (sometimes called zygotes) survive when a pond dries up. Bdelloid rotifers don’t produce resting eggs; they survive desiccation through a process called anhydrobiosis, contracting into an inert form and losing most of their body water. Embryos, juveniles and adults can undergo this process. The bdelloid withdraws its head and food and contracts its body into a compact shape called a tun; a generally unprotected dormant state that remains permeable to gases and liquids. Like Tardigrades (see below), Bdelloid rotifers can resist ionizing radiation because they can repair DNA double-strand breaks.
Tardigrades, also known as water bears or moss piglets, are plump, bilaterally symmetrical organisms with eight clawed legs. Fossils of tardigrades date to the Cambrian period over 500 million years ago. Over 900 species are known. Tardigrades were first described by the German pastor Johann August Ephraim Goeze in 1773 and given the name Tardigrada, meaning “slow stepper,” by the Italian biologist Lazzaro Spallanzani. Tardigrades reproduce asexually (parthenogenesis) or sexually. They mostly suck on the fluids of plant cells, animal cells, and bacteria. Tardigrades are, in turn, eaten by amoebas, nematodes, and other tardigrades. When their environment dries up, tardigrades (like bdelloid rotifers) shrivel up into a tun and wait it out.
Creative Survival
As I mentioned already, many of these invertebrates have evolved creative ways to survive in very unstable environments. Take a puddle or ephemeral pond, for instance. It dries up then reappears with rain. How can these communities thrive? Or do they all die off and then somehow recruit when the pond reappears?
In 1701, Antonie van Leeuwenhoek observed that “animalcules” (likely the bdelloid rotifer Philodina roseaola) survived desiccation and were “resurrected” when water was added to them. He’d discovered a highly resistant dormant state of an aquatic invertebrate to desiccation.
Dormancy is a common strategy of organisms that live in harsh and unstable environments and has been documented in crustaceans, rotifers, tardigrades, phytoplankton and ciliates. “Dormant forms of some planktonic invertebrates are among the most highly resistant … stages in the whole animal kingdom,” writes Jacek Radzikowski in a 2013 review in the Journal of Plankton Research. Radzikowski describes two states of dormancy: diapause and quiescence.
Diapausing results in the production of a dormant egg or cyst whose thick envelope or shell protects it from drying, freezing, mechanical damage, microbial invasion and predation, UV radiation and harmful chemicals. Many survive being eaten and can resist vertebrate digestive enzymes, helping them disperse and colonize isolated water bodies. Diapause is controlled by an internal mechanism that is initiated by various cues, such as temperature or photoperiod. In short-lived organisms, it is typically initiated only in a single ontogenetic stage. “Breaking of diapause requires specific cues, and not necessarily the return of favorable conditions,” writes Radzikowski.
Quiescent dormancy does not involve the production of a dormant egg or cyst; rather it involves a transformation of the organism itself into a dormant state through a process called cryptobiosis. “Quiescence is … induced directly by the occurrence of harsh environmental conditions. A quiescent organism can enter this state in many stages of its life, and remains dormant only until the adverse conditions end.”
Bdelloid rotifers can enter quiescent dormancy in the embryo, juvenile or adult stage of their lives. The dormant stages of copepods vary among the three orders. Many calanoid species can produce dormant eggs, but some calanoids can enter dormancy as nauplii or copepodids. Dormancy in cyclopoids is restricted to copepodids and adults, while the benthic harpacticoids can enter dormancy as eggs, nauplii, copepodids or adults. As they enter dormancy, the copepod stage lowers its metabolism, empties its gut and reduces movement of its intestine. Digestive enzymes reduce and lipid globules increase. The animal then goes rigid and colourless.
Cladocerans switch from eggs produced parthenogetically to sexually produced dormant eggs when conditions deteriorate. Copepod dormant eggs are produced bisexually. Ostracod dormant eggs can be produced either bisexually or through parthenogenesis. These dormant eggs survive desiccation through heat-shock proteins, such as disaccharides and glycerol, which act as cryoprotectants. Bdelloid rotifers and tardigrades use a similar mechanism during quiescent dormancy.
Extreme Behaviour & Bizarre Tolerances
Several aquatic invertebrates produce dormant forms that tolerate environmental extremes and stresses that they will never encounter in their natural habitats.
In 1940, Luyet and Gehenio reported that dried brine shrimp (Artemia salinas) cysts, taken from the Great Salt Lake in Utah, survived exposure to near absolute zero degrees Centigrade for six days and hatched successfully. Artemia cysts also resisted very high temperatures, and levels of desiccation not normally found on Earth (Clegg et al., 1978).
Bdelloid rotifers can go into quiescent dormancy at practically any stage in their life cycle in response to unfavorable conditions. Early research noted that dormant animals could withstand freezing and thawing from −40°C to 100°C and storage under vacuum. They also tolerated high doses of UV and X radiation. Later work reported that some rotifers could survive extreme abiotic conditions, such as exposure to liquid nitrogen (−196°C) for several weeks or liquid helium (−269°C) for several hours. Desiccated adult bdelloid rotifers apparently survived −80°C conditions for more than 6 years. The dormant eggs of cladocerans and ostracods also survived below freezing temperatures for years.
The Tardigrade
Tardigrades are the “poster children” for tolerating—if not outright thriving in—extreme environments. They can withstand the following:
- High and low temperatures (e.g., -273°C to 151°C; the lowest temperature ever recorded on Earth was -89.2°C in central Antarctica in 1983)
- freezing and thawing
- changes in salinity
- lack of oxygen
- lack of water
- levels of X-ray radiation 1000x the lethal human dose
- some toxic chemicals
- boiling alcohol
- low pressure of a vacuum
- high pressure (up to 6x the pressure of the deepest part of the ocean).
Tardigrades respond to adverse environmental stresses through a process called cryptobiosis, in which the organism arrests its metabolic activities without dying.
Anhydrobiosis—a form of cryptobiosis in response to lack of water—is commonly practiced by tardigrades. As with bdelloid rotifers, tardigrades survive dry periods by curling up into a little ball or tun. A protective sugar called trehalose is made and moves into the cells to replace the lost water. You could say that the water bear turns into a gummy bear. While in a tun, the tardigrade’s metabolism reduces to less than 0.01% of normal. Revival usually takes a few hours. Tardigrades have revived from dried moss kept in a museum for over 100 years when water was added. When a tardigrade starts to dry out, it makes lots of antioxidants—chemicals that, like vitamin C and E, soak up dangerously reactive chemicals. When a tardigrade is dried out for a long time, its DNA may get damaged; the antioxidants may help its ability to repair its DNA.
When the oxygen gets too low for the tardigrade to respire, it stretches out into a long, relaxed state with a highly reduced metabolism (in a process called anoxybiosis); the relaxed muscles allow as much water and oxygen to enter their cells. If the surrounding temperature plummets below freezing, the tardigrade forms a cold-resistant tun (called cryobiosis), preventing ice crystals that could damage cell membranes from forming. Tardigrades also practice osmobiosis in response to increased solute concentration, such as salt water.
Even though tardigrades can withstand many extreme environments, they’re not considered extremophiles, given that they don’t thrive in these conditions—as true extremophiles do.
Having said this, tardigrades survive adverse environmental stresses that do not naturally occur on Earth (and must be created in the lab). For instance, the lowest temperature ever recorded on Earth was -89.2°C in central Antarctica in 1983; yet a tardigrade can survive a freezing temperature of -273°C. Tardigrades can withstand ionizing radiation and pressures not found on Earth.
All this raises questions of origin, evolutionary adaptation, and niche partitioning and movement. How—and why—have tardigrades developed the ability to survive the vacuum and ionizing radiation of space? Amateur tardigrade enthusiast Mike Shaw postulated that these animals are equipped to survive in outer space because they originally came here from outer space (from some other planet supposedly). Scientists argue that tardigrades developed these extreme tolerances to outer space right here on Earth from particularly volatile environments (e.g., water bodies that freeze or dry up, and undergo periods of anoxia, etc.); the logical deduction is—if they can make it there, they can make it anywhere. So, which came first: the chicken or the egg? Where is “home”, really?…

Nina Munteanu is an ecologist, limnologist and internationally published author of award-nominated speculative novels, short stories and non-fiction. She is co-editor of Europa SF and currently teaches writing courses at George Brown College and the University of Toronto. Visit www.ninamunteanu.ca for the latest on her books.












Hi,
we would very much like to use on of your beautiful photographs in a book we are about to publish. Could you contact me under the email address I entered?
Thank you very much,
T
LikeLike
Hi Tim,
Contact me and we can discuss. All the Best,
Nina
LikeLike