
Nina inspecting a rock for benthos of Credit River, ON (photo by Merridy Cox)
During the years when I taught Limnology and Aquatic Ecology at the University of Victoria in British Columbia, one of the first things I did was take my students on a field trip to several urban streams and ponds. Usually one or two students came prepared to go into the water, bringing the requisite equipment like boots or old sneakers to change into. I would “volunteer” them to wade into the stream to examine and dislodge a few rocks for the others to see.

Nina in gleeful discovery (photo by Merridy Cox)
It was so fun to witness the startled glee in my students’ faces when they turned the rocks over to discover the active life that lurked there. It was one thing to know that benthic (bottom-dwelling) life clung to surfaces; it was quite another to be confronted with the reality of this rather bizarre community. My student Florence could hardly hold onto the rock as stunned mayflies and stoneflies scurried for cover. Glossoma caddisflies peered out of stone “houses” glued to the rock, no doubt hoping for reinstatement in the cool, dark streambed—which my student kindly obliged them. Once upon a time, I was Florence, awestruck by Nature’s hidden surprises…
I began my limnology career as a stream ecologist and the focus of my master’s study included several rural and urban streams in the Eastern Townships of Quebec, Canada, not far from where I grew up. Later, as a limnologist for various environmental consulting companies in British Columbia, I studied the nature of macro-benthos communities in streams impacted by various activities from industrial discharges to agriculture and municipal development.
Macro-benthos is bottom-dwelling life you can see with the naked eye. They’re made up mostly of aquatic worms and juvenile stages of insect species (benthic invertebrates).
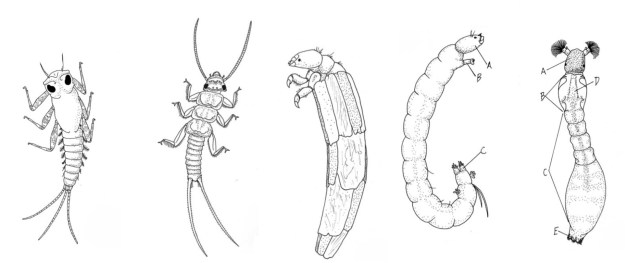
From left to right, Mayfly, Stonefly, Caddisfly, Chironomid, Blackfly larva (illustrations by Kerste Voute, in “Water Is…The Meaning of Water”)
Stream ecologists identify benthic invertebrates by their form, but they also recognize them by how they feed. Shredders, such as amphipods, mayflies, stoneflies, midges, and some caddisflies, use scissor-like mouths to cut and shred apart coarse particulate matter such as leaves and other plant material. Collector–gatherers (e.g., worms, nematodes, crustaceans, mayflies and gastropods) use their broom-like mouths to sweep in fine and ultra-fine organic matter that includes feces of shredders. Filtering collectors, such as black fly larvae, collect suspended fine and ultra-fine organic matter, which can include phytoplankton (floating algae) in their finely spun nets.

Net-spinning Hydropsychidae caddisfly larva, showing the window-screen net at the mouth of its tube (photo by Merridy Cox)
The caddisfly larva Arctopsychegrandis, which belongs to the Hydropsychidae, builds a rough house made from twigs, leaf fragments, and small pebbles and spins silk nets across its threshold to capture organic matter suspended in the stream that includes algae, detritus, and smaller invertebrates.
 Grazers or scrapers, such as beetles, mayflies, and stoneflies, feed on attached algae and biofilms. Their mouths chisel against periphyton (attached algae) on rocks, woody debris and aquatic plants. Filtering collectors, such as black fly larvae, collect suspended fine and ultra-fine organic matter, which can include phytoplankton (floating algae) in their finely spun nets. Predatory benthic invertebrates, such as larval damselflies and dragonflies, have piercing mouth parts that act like a straw, allowing them to suck the nutrients from their prey—other invertebrates—without having to chew or shred it.
Grazers or scrapers, such as beetles, mayflies, and stoneflies, feed on attached algae and biofilms. Their mouths chisel against periphyton (attached algae) on rocks, woody debris and aquatic plants. Filtering collectors, such as black fly larvae, collect suspended fine and ultra-fine organic matter, which can include phytoplankton (floating algae) in their finely spun nets. Predatory benthic invertebrates, such as larval damselflies and dragonflies, have piercing mouth parts that act like a straw, allowing them to suck the nutrients from their prey—other invertebrates—without having to chew or shred it.
In his article “Caddisflies: Architects Underwater” in the Summer 2007 Issue of American Entomologist, Glenn B. Wiggens shows a histogram of the number of species from each of the three main orders of aquatic insects (Ephemeroptera, Plecoptera, and Trichoptera) that are shredders, collectors, scrapers and predators. Mayflies (Ephemeroptera) are mainly collectors and scrapers; stoneflies (Plecopera) are mainly predators and shredders; caddisflies (Tricoptera) show a more even distribution of feeding strategies.
Trent Nature Sanctuary Walk

Beaver activity in forest stream, ON (photo by Nina Munteanu)
I recently went walking with naturalist Merridy Cox along some trails in the Trent Nature Sanctuary forest of cedar-hemlock-maple-beech-birch. The sanctuary occupies the southern-most part of the Great Lakes-St. Lawrence forest region, dominated by mixed forest that features hardwoods such as maple, oak, yellow birch, poplar and conifers such as white and red pine, hemlock and white cedar.
Aside from enjoying the complex forest ecosystem, I was always on the look out for water: ponds, marshes, swamps and streams. I saw examples of all of these.
KnowledgeNuts defines marshes and swamps this way: “Marshes are nutrient-rich wetlands that support a variety of reeds and grasses, while swamps are defined by their ability to support woody plants and trees.” Both swamps and marshes are transitional areas between land and an aquatic ecosystem. These hybrid regions of two ecosystems are called ecotones.
Beaver Marsh in the Making…

Beaver-topped tree (photo by Nina Munteanu)
My first example came on the west Red Trail, where beaver activity was starting to create an open marsh along a forest stream. The trail’s boardwalk meandered through a soggy area of reeds, grasses, red osier dogwood, marsh marigolds, and drowned trees—mostly cedar, yellow birch and poplar. Several trees—mostly poplar—had been taken down by the busy beavers. Sedges, rushes such as cattails, grasses and ferns were pushing up their spring growth amid the dead winter vegetation. I wondered if the beavers would successfully build their dam and if the area would eventually turn into an actual pond.
Swamp Forest (Palustrine wetland)

Moss-covered stump in forest swamp, ON (photo by Nina Munteanu)
Along the east Red Trail, Merridy and I were following a stream along a path when I noticed a lowland area of flooded forest. I pushed my way through the young cedars and found myself in a swamp of trees standing in a spongy water-logged black mat of decaying leaves, moss islands, logs and large debris. There was standing water farther into the swamp of mostly silver maple trees with some green ash and swamp oak. We also noted some elm, birch, alder and poplar trees amid the cedars that lined the swamp. This waterlogged area is high in nutrient content and these trees have adapted to the wetness that would rot the roots of other trees. However, because of their shallow roots, blow-downs often occur, creating openings in the forest canopy, which brings opportunity for more growth. Numerous logs colonized by thick moss quilted the swamp with vivid green. The pit and mound topography provides a diverse habitat for a wide range of species.

Forest swamp in Trent Nature Sanctuary, ON (photo by Nina Munteanu)
According to Nature Conservancy Canada, these forest swamps are so productive that they are often compared to rain forests and coral reefs. Swamp forests provide a variety of ecological function, including: slowing drainage flows from developed areas; reducing floods; filtering out pollutants; and trapping sediments.
The water levels in these swamps fluctuate with the season and because of its unique topography and hydrology, the forest swamp provides habitat for a number of significant species such as the prothonotary warbler—endangered by loss of its habitat. I will be returning to see if I can spot this rare bird.
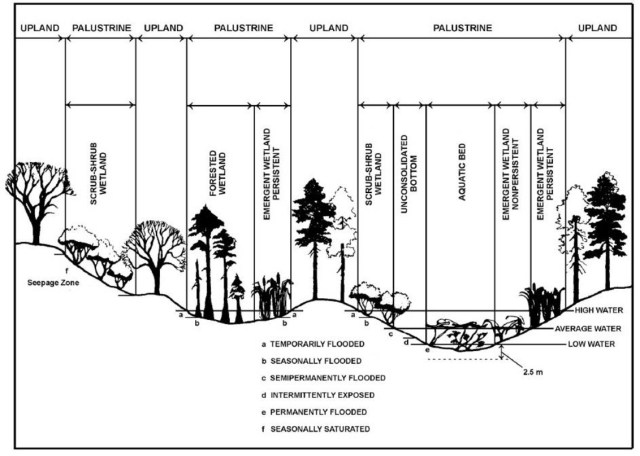
Palustrine wetland (USGS)
Vernal Pool
Our walk in the forest took us on several boardwalks over wetlands and past vernal pools—temporary ponds of standing water full of forest litter: decaying leaves from the surrounding trees, debris and other organic material. Don Scallen of In The Hills tells us that “vernal pools evaporate in summer’s heat, but during their temporary presence they are factories of biodiversity, nurseries for some of the most beautiful and important woodland creatures.”

Vernal pool in Trent Nature Sanctuary forest, ON (photo by Nina Munteanu)
When I stopped to peer more closely into the pool, I saw that many of the twigs and bits of leaves were moving. I instantly recognized them as case-building caddisflies. the majority were of the same species of plant-building caddisfly: Limnephilus externus.

Limnephilus externus caddisflies foraging on decaying leaves of a vernal pool (photo by Nina Munteanu)
I had previously seen rock and sand-building caddisflies and net-spinning caddisflies in streams and lakes. I’d also seen several cases built in long narrow tubes of intricately placed leaves in detritus-filled shallows. However, these thick log-houses with material arranged transversely rather than lengthwise, were a new find for me. And this vernal pool was teaming with them. Thousands of Limnephilus milled through the decaying leaves, colliding into one another like a bumper car rally, and happily shredding up the decaying leaves and detritus.

Limnephilus externus case (photo by Merridy Cox)
The caddisflies built their chunky log cabins from a variety of leaves, twigs, stems, seeds, moss and other materials—whatever was around them—that they wrapped around themselves to protect against predators such as aquatic beetles, bugs and dragonfly nymphs.
Scallen writes that “there are dragonfly larvae with alien-like extendable mandibles, sickle-jawed diving beetle larvae three centimetres long or more, as well as what may be Canada’s most fearsome invertebrate predaors—giant water bugs that wield piercing beaks to drain the bodily fluids of prey as large as adult frogs.”
Fungi and bacteria that grow on the decomposing material also provide nutrients for the caddisfly. The case serves as protection and camouflage for the larva as it shreds through the leaf detritus. The by-product of shredding creates fine organic particles for filter-feeding and other collector larvae—including mosquitoes, midges, mayflies and crustaceans.

Vernal pool (photo by Nina Munteanu)
Vernal pools normally appear after snow melt in March and April and often don’t last beyond July. “Few species of aquatic insects of any kind can survive the alternating wet and dry phases of temporary pools,” writes Wiggins. “But every one that can live under these conditions has accrued a set of remarkable survival strategies through natural selection.” Along with a myriad of other aquatic life in the pool—I make out ostracods, copepods, daphnia, water shrimps, beetles and other insects—the caddisflies in this temporary pool have adapted over millennia to live in a short-lived wet habitat. One advantage is the lack of fish predation (native fish to this region cannot survive the dry phase and therefore don’t exist in temporary pools). A second advantage lies in the highly nutritious nature of decomposing plant detritus, enhanced by the temporary nature of flooding and drying. Colonizing fungi contribute much of the protein of decomposing plant materials. Adult Limnephilidae emerge from the pupal state as the pool recedes in June or July. After diapause and only once the days shorten in late summer, female adult caddisflies lay eggs on the underside of moist logs of the basin in a gelatinous matrix that resists desiccation for months. The tiny larvae leave the gelatinous matrix when the basin floods—usually after snow melts in the spring. Then the larvae crawl out, construct a case and start eating what’s around them.
As I watched the Limnephilus caddisfly larvae mill around the rotting leaves in the shallows of what looked like a busy intersection in New Delhi, I noticed the great diversity of their houses. Some were much fatter than others. Some were unevenly built; others were constructed with a meticulous sense of symmetry. Yet others appeared outlandish and flamboyant with one stem or cedar leaf resembling a flag. Yet another chose to give itself a mossy tail. It was as though I was witnessing diverse personalities being expressed in these individual cases. Not one was the same. Each was a unique arrangement of available materials but also of selective taste.

Limnephilus cases vary greatly in dimensions and materials (photos by Nina Munteanu)
One site in Colorado claimed that the case structure, while obviously varying by available material, was also triggered by presence or absence of predators and the density of other Limnephilus externus. They claimed that when food supplies were low, the Limnephilus would chew each other’s cases down. Imagine someone coming to your house and eating away your second bedroom! Wissinger et al. partially supported this claim by showing in the lab that Limnephilus supplemented its detrital diet with animal material, which included preying on other caddisflies. The researchers concluded that the larval case reduced aggression and cannibalism in temporary wetlands.
Caddisfly cases made of stone, sand, twigs, leaves and other materials
According to Environment and Climate Change Canada estimates, “about 70 to 85 per cent of wetlands in southern Ontario have been lost over the past century. It’s hard to assess the loss of vernal pools because they are small and often hidden among trees, but it is likely that in the near-urban countryside most are gone. Forests that harbour vernal pools have been cleared, and much of the landscape has been drained and levelled for agriculture and urban development. It is sobering to think an hour or two of earth moving is all it takes to fill in a vernal pool – and that this brief act of ecological vandalism can diminish local biodiversity forever.”–Don Scallen, In the Hills
 In Piotr Naskrecki’s wonderful book The Smaller Majority, the entomologist, photographer, author and Harvard professor advocates passionately for the conservation of invertebrates and other small creatures so important to the health of our planet.
In Piotr Naskrecki’s wonderful book The Smaller Majority, the entomologist, photographer, author and Harvard professor advocates passionately for the conservation of invertebrates and other small creatures so important to the health of our planet.
Naskrecki writes, “Public appreciation of the beauty and importance of these animals is our strongest ally in this conservation work.” He encourages us to “kneel down, look closer and discover the beautiful world around our feet.”
Though no equipment other than warm clothes is necessary, your experience will be enhanced by taking rubber boots, dip net, camera, magnifying lens, small plastic container for viewing animals and close-focus binoculars.

Nina Munteanu exploring Thompson Creek, ON
Ontario Nature, a province-wide conservation organization, runs a vernal pool mapping project to document the location of vernal pools. The objective is to identify priority areas for research and conservation efforts. If you find a vernal pool, you can report it on the Vernal Pool Search form at ontarionature.org.
Much of this article is excerpted from “Water Is…The Meaning of Water” (Pixl Press).

References:

Marshy stream impacted by beaver activity (photo by Nina Munteanu)
Allan, D.J. 2007. “Stream Ecology: Structure and Function of Running Waters.” 2nd edition. Springer. 436 pp.
Cummins, K.W. and M.J. Klug. 1979. “Feeding Ecology of Stream Invertebrates.” Ann.Rev. Ecol. Syst. 10: 147–172.
Merritt, Richard W. and Kenneth W. Cummings. 1996. “An Introduction to the Aquatic Insects of North America.” Kendall/Hunt Publishing, Dubuque. 862 pp.
Munteanu, N. 2006. “Fitness Indicators and Morphological Deformities of Benthic Invertebrates as an Assessment Tool in Ecosystem Health.” In: Benthic Biomonitoring July 25–26, 2006. British Columbia Ministry of Environment and the University of British Columbia, B.C.
Munteanu, N. 2016. “Water Is…The Meaning of Water.” Pixl Press, Vancouver. 585pp.
Munteanu, N. and G.P. Thomas. 1997. “Benthic Community Populations Near Two Adjacent Northern Pulp Mill Discharges.” Water Sciences & Technology 35 (2/3): 381.
Scallen, Don. 2017. “Vernal Pools.” In the Hills. Online: https://www.inthehills.ca/2017/03/vernal-pools/
Thomas, G.P. and N. Munteanu. 2000. “Biological Community Assessment Program; Benthic Invertebrate and Periphyton Communities of the Columbia River.” Report prepared for Cominco Canada Ltd. by G3 Consulting Ltd., Richmond, BC.
Wiggins, Glenn B. 2015. “Caddisflies: The Underwater Architects.” The University of Toronto Press, Toronto, ON. 300pp.
Wiggins, Glenn B. 2007. “Caddisflies: Architects Under Water.” American Entomologist 53 (2): 78-85.
Wissinger, Scott A., Charles Eldermire, and John C. Whissel. 2004. “The role of larval cases in reducing aggression and cannibalism among caddisflies in temporary wetlands.” Wetlands 24 (4): 777-783.

Beaver-mediated stream and wetland in Trent Nature Sanctuary, ON (photo by Nina Munteanu)

Nina Munteanu is a Canadian ecologist / limnologist and novelist. She is co-editor of Europa SF and currently teaches writing courses at George Brown College and the University of Toronto. Visit www.ninamunteanu.ca for the latest on her books. Nina’s bilingual “La natura dell’acqua / The Way of Water” was published by Mincione Edizioni in Rome. Her non-fiction book “Water Is…” by Pixl Press(Vancouver) was selected by Margaret Atwood in the New York Times‘Year in Reading’ and was chosen as the 2017 Summer Read by Water Canada. Her novel “A Diary in the Age of Water” will be released by Inanna Publications (Toronto) in June 2020.

A walk through a forest with ecologist/limnologist Nina Munteanu reveals all sorts of wonders and surprises after every turn in the trail.
LikeLiked by 1 person
Thanks, Merridy! Those walks in the forest are real adventures of exploration and discovery, aren’t they?
LikeLike