
A while ago, when I was a student at Concordia University, I studied diatoms in freshwater streams in the Eastern Townships for my Master of Science degree. Diatoms are microscopic plantlike organisms that make a hard silica (glass) cell wall (called a frustule) that is often beautifully marked and striated. It is these markings that phycologists use to identify the many diatom species that exist.

Diatoms have often been called “jewels of the sea” and “living opals” because of their photonic nanostructure. Diatomaceous earth is used in a variety of things from water filtration, as a mild abrasive (e.g., in toothpaste), and in cat litter. Diatoms live in oceans, freshwater and soils and number in the trillions. They generate some 20 percent of the oxygen produced on the planet and contribute nearly half of the organic material found in the oceans.
During my research in several streams in the Eastern Townships, I examined how diatoms colonized artificial substrates; how they formed productive biofilms that sustained an entire periphyton community of attached aquatic life. I submerged glass slides (the kind people use to look at critters under the microscope) in a device in the stream and oriented them parallel to the current.

There are two ways an algal community grows in a new area: (1) by initial colonization and settling; and (2) by reproduction and growth. I studied both by collecting slides exposed for differing lengths of time (collecting young and mature communities) in different seasons.
I discovered that the diatoms colonized these surfaces in weird ways based on micro-turbulence. Early colonizers, like Achnanthes and adnate Cocconeis preferred to settle on the edges of the slides, where the chaos of turbulence ruled over the sheer of laminar flow. They colonized by directly appressing to the substrate, making them the first photosynthetic taxa to establish a biofilm on a clean substrate. Vadeboncoeur and Katona (2022) write that “in waved-washed surfaces, these taxa may be the only algae that persist.” I postulated that the drift velocity was reduced on the slide’s edge, where turbulence was greatest, giving drifting algae a greater chance to collide and settle on the slide over the more shear laminar flow along the slide’s central face.
Once settled, the community was more likely to grow with turbulence. Greater turbulence decreases the diffusion gradient of materials around algal cells, with a higher rate of nutrient uptake and respiration. Turbulence provides greater opportunity to an existing colony by increasing “chaotic” flow, potential collision and exchange. Turbulence is a kind of “stable chaos” that enhances vigor, robustness and communication.
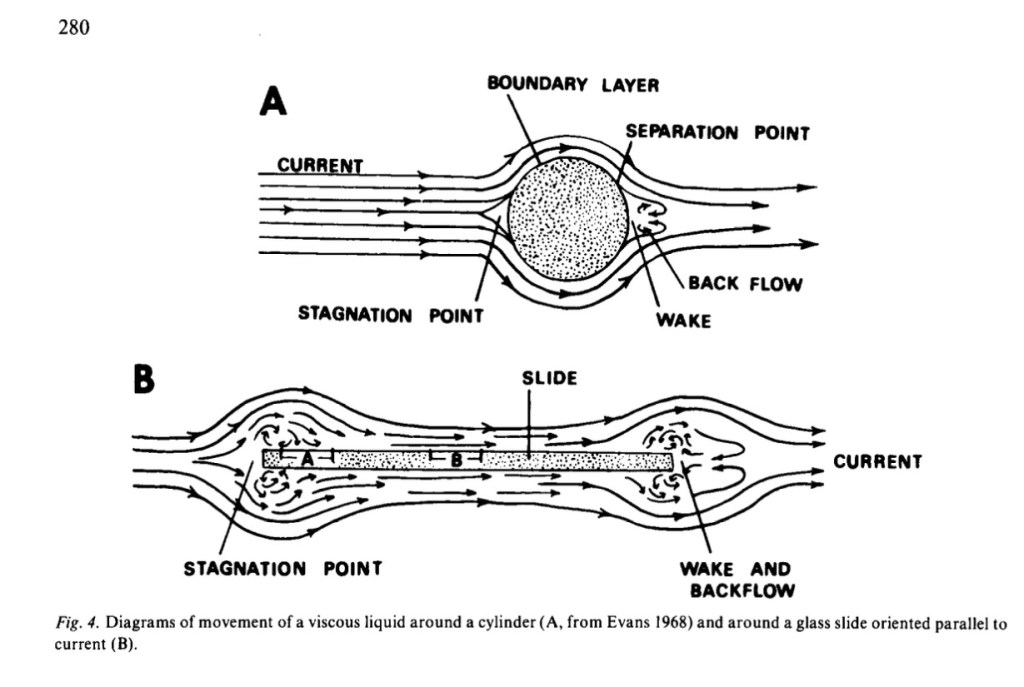
Without quite realizing it, I’d discovered one of nature’s most intriguing and fundamental qualities and a tenet in ecology: at the boundary, where two different entities meet—liquid with solid, land with water, forest with field, freshwater with salt water, fast water with slow water—life thrives most vigorously.
Ecologists have a name for this zone: they call it an ecotone. At the root of this vitality lies movement: turbulence, spiraling, swirling, vortices, flushing, and, yes, collision and excitation, exchange, learning, adaptation, change, and evolution.
My paper was cited by many other studies and the term “edge effect” came into the scientific lexicon. And speaking of edges, let’s talk about forests and how they come about…

How Diatoms Create a Forest…
Think of how a terrestrial forest forms through succession after a fire or other clearing (first with mosses, forbs, graminoids and shrubs, then young saplings of pioneer trees establishing understory and eventually overstory with a mature tree canopy); that’s very much how a diatom forest forms. The key is attachment: diatoms adhere to submerged surfaces through mucilage produced between the frustule (the diatom’s outer shell) and its substrate. The form of the mucilage varies and includes gelatinous stalks, pads and tubes.
Attachment and colonization starts with a ‘clean’ unpopulated surface (usually scoured by turbulence in a storm or some other event or a new surface tumbled into the water). Several stages of succession take place, starting with early colonizers. The adnate Cocconeis placentula, whose frustules attach directly to the substrate, is an example of an early colonizer (think overstory and understory of a terrestrial forest or a marine kelp forest); adnate diatoms like Cocconeis are first to attach to a substrate and form a biofilm (think moss in a terrestrial forest). Adnate species are eventually overgrown by taxa that produce a mucilaginous pad (e.g. Synedra) or stalk (e.g. Gomphonema) that allows them to protrude above the adnate taxa and take advantage of more light.
As part of the understory layer, diatoms such as Fragilaria vaucheriae and Synedra radians attach to the surface at one end (apical) of their rod-shaped frustules using a mucilaginous pad to form “rosettes” that resemble spiky understory shrubs.

Cymbella and Gomphonema produce long gelatinous stalks that attach directly to the surface, allowing them to form a swaying canopy over the lower tier of cells such as Fragilaria vaucheriae, Synedra radians.
Just like trees, the canopy-forming stalked diatoms effectively compete for available light and nutrients in the water with their vertical reach. They provide the ‘overstory’ of the diatom forest’s vertical stratification. These tree-like diatoms also provide an additional surface for other diatoms to colonize (e.g. tiny epiphytic Achnanthes settle on the long stalks of Cymbella, just as lichen does on a tree trunk).

The stalked diatom forest acts like a net, trapping drifting-in euplankton, such as Pediastrum sp. and Fragilaria spp., which then decide to stay and settle in with the periphyton community. Among them are the mobile, raphid taxa (e.g. Navicula) that glide up and down in pursuit of good light. The mucilage captures and binds detrital particles in both lower and upper stories of the diatom forest; these, in turn, provide nutrients for the diatom forest and additional surfaces for colonization by other organisms. In their work with periphyton communities, Roemer et al. (1984) found several diatoms (e.g. Diatoma vulgare, Fragilaria spp. Stephanodiscus minutula) entangled in the complex network of cells, stalks, and detritus of the diatom forest’s upper story. They also found rosettes of Synedra radians—like jungle orchids—attached to large clumps of sediment caught by the net of mucilage.
The diatom forest may also attract dense clouds of unattached filamentous green algae (e.g. Zygnema, Spirogyra, Mougeotia) or organic-loving blue green algae (e.g. Lyngbya, Oscillatoria).

Eventually, ‘overgrowth’ occurs as the periphyton colony matures and grows ‘top-heavy’ with all this networking. The upper story of the community simply sloughs off—usually triggered by turbulence in a river from rains, storms, or dam release. This is similar to a forest fire in the Boreal forest, which creates space and light for new colonization and growth. The dislodged periphyton ride the turbulent flow, temporarily becoming plankton, and those that survive the crashing waters provide “seed” to colonize substrates downstream. Others may get damaged and form the ‘dish soap’ suds or foam you often see in turbulent water. The proteins, lignins and lipids of the diatoms (and other associated algae) act as surfactants or foaming agents that trap air and form bubbles that stick to each other through surface tension.
To read my entire 1981 paper (“The effect of current on the distribution of diatoms settling on submerged glass slides” Hydrobiologia 78: 273-282) go to the Springer site and download.
References:
Lauterwasser, Alexander. 2006. “Water Sound Images: The Creative Music of the Universe.” Macromedia Publishing. 172 pp.
Diatoms of North America. 2021.
Munteanu, N. & R. Maly. 1981. “The effect of current on the distribution of diatoms settling on submerged glass slides.Hydrobiologia 78: 273-282.
Munteanu, Nina. 2016. “Water Is…The Meaning of Water.” Pixl Press, Delta, BC. 584 pp.
Palmer, Marvin C. 1959. “An Illustrated Manual on the Identification, Significance, and Control of Algae in Water Supplies.” U.S. Department of Health, Education, and Welfare, Cincinnati, Ohio. 98pp.
Roemer, Stephen C., Kyle D. Hoagland, and James R. Rosowski. 1984. “Development of a freshwater periphyton community as influenced by diatom mucilages.” Can. J. Bot. 62: 1799-1813.
Ross, R., Cox, E.J., Karayeva, N.I., Mann, D.G., Paddock, T.B.B., Simonsen, R. and Sims, P.A. 1979. “An amended terminology for the siliceous components of the diatom cell.Nova Hedwigia, Beihefte 64: 513-533.
Round, F.E., Crawford, R.M. and Mann, D.G. 1990. “The Diatoms. Biology and Morphology of the Genera.” Cambridge University Press, Cambridge. 747pp.
Schwenk, Theodor. 1996. “Sensitive Chaos.” Rudolf Steiner Press, London. 232 pp.
Smolar-Zvanut, Natasa and Matjaz Mikos. “The impact of flow regulation by hydropower dams on the periphyton community in the Soca River, Slovenia. Hydrological Sciences Journal 59 (5): 1032-1045.
Watson, Susan and Friedrich Jüttner. 2019. “Biological production of taste and odour compounds.” In: “Taste and Odour in Source and Drinking Water: Causes, Controls, and Consequences”, Chapter 3. IWA Publishing 63-112pp.
Wilkens, Andreas, Michael Jacobi, Wolfram Schwenk. 2005. “Understanding Water”. Floris Books, Edinburgh. 107 pp.
Wood, Allison R. 2016. “Attached Algae as an Indicator of Water Quality: A Study of the Viability of Genomic Taxanomic Methods.” Honors Theses and Capstones. 306. University of New Hampshire Scholars’ Repository.


Nina Munteanu is a Canadian ecologist / limnologist and novelist. She is co-editor of Europa SF and currently teaches writing courses at George Brown College and the University of Toronto. Visit www.ninamunteanu.ca for the latest on her books. Nina’s bilingual “La natura dell’acqua / The Way of Water” was published by Mincione Edizioni in Rome. Her non-fiction book “Water Is…” by Pixl Press (Vancouver) was selected by Margaret Atwood in the New York Times ‘Year in Reading’ and was chosen as the 2017 Summer Read by Water Canada. Her novel “A Diary in the Age of Water” was released by Inanna Publications (Toronto) in June 2020.







It’s interesting to understand what is happening within the waterbodies nearby as we walk along a pathway.
LikeLiked by 1 person
Yes, it certainly is, Merridy. There is so much hidden to the regular eye, a whole world of worlds!
LikeLike