On a late July morning, I was on my daily walk along the Otonabee River in the Kawarthas when I noticed a yellow-brown creamy film on the water lapping onshore. In some places it looked like left-over dishwater; in other places where it had a better chance to collect, it resembled cream. The multi-hued film swirled like flowing paint in an Emma Lindstrom artwork. It was the “eddy shedding” patterns of Saturn’s north polar atmosphere or Jupiter’s Great Red Spot.
Otherworldly. But it didn’t end there…
Later in the afternoon Environment Canada issued air quality warnings as smoke plumes filled with particulates from active fires in northwestern Ontario had settled into the area. The air indeed smelled like a campfire and the sun started to look more like a flaming copper penny.
The froth had turned into a mocha-coloured micro-foam that resembled the crema of a well-made espresso. Entrained by the wind that conspired with the river’s current, the film grew into caramel ropes that covered the rocks on shore in stripy shades of delicious earthy mocha cream—a synesthete’s dream.
Was this literally smoke on the water?




At the mouth of one little tributary to the river, foam with hints of caramel rose in a cloud of froth where the runoff tumbled into the Otonabee River.
Dust and Diatoms
I thought of the smoky day.
In spring, pollen creates films of yellow-green particles on water. Why not smoke? The large and small airborne particles from burnt trees would eventually fall from the air as the negatively charged ions created by moving water attached to the positively charged dust; the dust then becomes too heavy to stay airborne and falls into the water. Negative ions are basically oxygen ions with an extra electron attached, produced through water molecules—particularly when water is agitated through movement and turbulence. You can read more about this in my book Water Is… The Meaning of Water.
I’d often seen foam on moving water, the kind caused by crushed diatoms (single-celled algae that live in rivers and lakes). The foam is often tinged with brown, sometimes creamy-coloured, but typically has larger bubbles and a dishsoap consistency. Diatom foam is a regular occurrence during the lifecycle of diatoms, which can make up a significant portion of river biota.
I had to admit, this was foam with attitude; this was designer foam, café crema micro-foam. I figured that the increased dust from smoke particulates conspired with the normal actions of river water on the diatom community to create something new—Dust and Diatoms.
Diatoms are enclosed in shells made of nanopatterned silica and organic compounds called frustules. The glass frustules are hard but porous and etched in decorative markings or bands (rows of pores or alveoli that form striae and interstriae) that are used in identifying the more than 20,000 species. The silica shell protects the diatom from predators and acts as a ballast; the pores give the diatom access to the external environment for things like nutrient uptake, waste removal, and mucilage secretion so the diatom can attach itself to a substrate.

When subjected to great turbulence—such as when the dams along the river suddenly open—the diatoms, along with the fatty acids of associated decaying organic matter, create a ‘soap’ as their glass shells break apart and release lipids and proteins that act as foaming agents or surfactants. Air bubbles are pulled in through the turbulence to produce foam. Because the organic surfactants lower the surface tension of water, the bubbles persist at the water’s surface. The bubbles accumulate hydrophobic substances and the dissolved organic matter stabilizes them and aggregates them as nutrient-dense foam.
The addition of copious amounts of tiny particles from excessive smoke and dust in the air—pulled into the water by negative ions—changes the consistency, colour, and behaviour of the foam.
Like I said: café crema.
This metaphor isn’t farfetched: in fact, crema—the most prized aspect of a well-made espresso—is created through a similar process when hot water emulsifies coffee bean oils and floats atop the espresso with smooth little bubbles.
Given that the Otonabee River is regulated by several hydro-generating dams and locks to control its flow and provide electricity, it’s not surprising that many of the diatoms in the foam are periphyton (attached algae) that have sloughed off a submerged surface in the sudden turbulence caused by the dam release. You’ve seen periphyton; they are the slippery brownish, oily-looking felt mats that cover the cobbles and rocks of streams and rivers. Sometimes, they grow bright greenish ‘hair’, most likely one of the three most common filamentous green algae (Spirogyra, Zygnema, and Mougeotia), all of which I’d already observed growing on the river banks in the spring.
Periphytic diatoms attach to surfaces in a number of ways: directly on their valve face (adnate); by stalks or pads (pedunculate); through pole attachment (rosettes. stellate or zigzag forms); or within a mucilaginous tube, itself attached to the substrate. Many diatoms are either tube-dwelling diatoms (e.g. Cymbella, Navicula) or diatoms growing on mucilaginous stalks (e.g. Gomphonema, Gomphoneis).

Tube-dwellers and stalked diatoms tend to dominate benthic environments of altered hydrological and thermal regimes downstream of dams. These mucopolysaccharide materials also ‘glue’ everything together into a dense micro-foam.

When the Forest Fire Meets the Diatom Forest (The Forest Under the Microscope)
Excited to see if I was right in my initial assessment, I returned with a small glass jar and sampled some of the tan-brown scum-froth then returned home to the microscope.
I was right; most of the scum was a combination of:
- detritus (decaying organic material from organisms and likely smoke particles from the northern forest fire),
- living diatoms and other algae, and
- the remains of many broken and empty diatom frustules
Most of the diatoms, other algae and microorganisms that I observed in the frothy scum are common periphyton: algae and associated organisms that cling to substrates such as rocks, sand, plants and, of course, reservoir walls. The periphyton ecosystem—like a terrestrial forest ecosystem—is three dimensional, consisting of layers upon layers of organisms, each adapted to its little niche in the “diatom forest.”
Diatoms naturally contribute to foam observed in rivers as they grow, die and break open. This time—in late July on the weekend of air quality warnings amid a distinct campfire smell and blushing sun from the smoke of a northern forest fire—the diatom foam took on a different quality, the cafe crema look of dust and diatoms, when the northern forest fires meet the diatom forest.
Looking at the Vertical Stratification of a Diatom Forest
Diatoms adhere to submerged surfaces through mucilage produced between the frustule (the diatom’s outer shell) and its substrate. The form of the mucilage varies and includes gelatinous stalks, pads and tubes. For instance, Cymbella and Gomphonema produce long stalks that attach directly to the surface, allowing them to form a swaying canopy over the lower tier of cells that attach flat or perpendicular to the surface such as Fragilaria vaucheriae, Synedra radians and Cocconeis placentula (think overstory and understory of a terrestrial forest or a marine kelp forest).

As part of the understory layer, diatoms such as Fragilaria vaucheriae and Synedra radians attach to the surface at one end (apical) of their rod-shaped frustules using a mucilaginous pad to form “rosettes” that resemble spiky understory shrubs.
Just like trees, the canopy-forming long-stalked diatoms effectively compete for available light and nutrients in the water with their vertical reach. They provide the ‘overstory’ of the diatom forest’s vertical stratification. These tree-like diatoms also provide an additional surface for other diatoms to colonize (e.g. tiny Achnanthes settle on the long stalks of Cymbella, just as lichen does on a tree trunk).
The stalked diatom forest acts like a net, trapping drifted-in euplankton, such as Pediastrum sp. and Fragilaria spp., which then decide to stay and settle in with the periphyton community. The mucilage captures and binds detrital particles in both lower and upper stories of the diatom forest. These, in turn, provide nutrients for the diatom forest and additional surfaces for colonization. In their work with periphyton communities, Roemer et al. (1984) found several diatoms (e.g. Diatoma vulgare, Fragilaria spp. Stephanodiscus minutula) entangled in the complex network of cells, stalks, and detritus of the diatom forest’s upper story. They also found rosettes of Synedra radians—like jungle orchids—attached to large clumps of sediment caught by the net of mucilage.
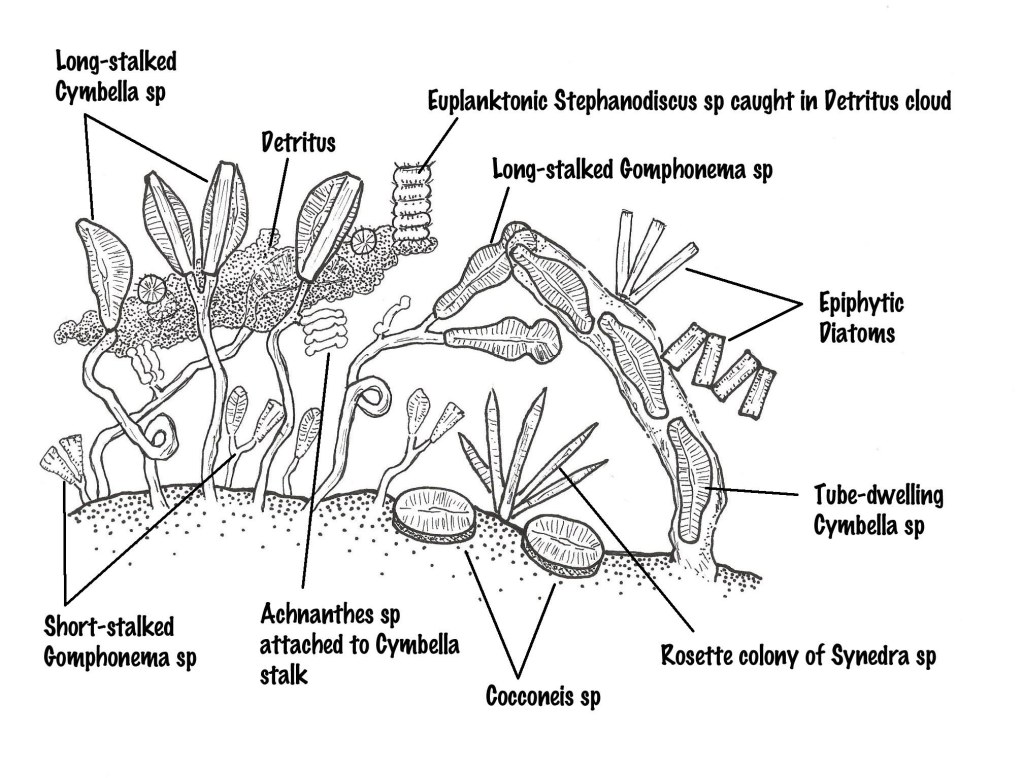
Eventually, ‘overgrowth’ occurs as the periphyton colony matures and grows ‘top-heavy’ with all this networking. The upper story of the community simply sloughs off—usually triggered by turbulence such as when the dams release water. This is similar to a forest fire in the Boreal forest, which creates space and light for new colonization and growth. The dislodged periphyton then ride the turbulent flow, temporarily becoming plankton, and those that survive the crashing waters provide “seed” to colonize substrates downstream.
When observing my scum under the microscope I saw many dead (empty) and broken diatom frustules in the detritus; I also observed many living diatoms along with green and blue-green algae, rotifers, Protista and bacteria feeding on the detritus. I even saw one amoeba, actively feeding on the nutrient-rich interface of a micro-foam organic bubble. I was unable to identify and separate smoke particles of the northern forest burn from the detritus normally created by the river itself, but there was a lot of detrital material there.
The table below provides a list of the periphyton and planktonic organisms that I identified in the scum after a few hours of random microscope scans on several subsamples magnified at x100 and x400.
Organisms Found in Otonabee River Surface Scum Downstram of Stanley Adamson Generating Station and Between Two Downstream Dams, July 2021
| Organism | Type | Frequency | Habitat / Designation1 |
| Cymbella spp. | Tube-dwelling / stalked Diatom | Common | Reservoir walls; Filter-clogging |
| Cocconeis sp. | Diatom | Common | Reservoir walls; clean water |
| Achnanthes sp. | Stalked Diatom | Common | Reservoir walls |
| Gomphonema spp. | Stalked Diatom | Common | Reservoir walls; polluted water; Eutrophic |
| Gomphoneis sp. | Stalked Diatom | Several | Periphyton |
| Diatoma sp. | Diatom | Several | Filter-clogging |
| Navicula sp. | Stalked Diatom | Several | Clean water; Filter-clogging |
| Stauroneis sp. | Diatom | Several | Plankton / Periphyton |
| Fragilaria sp. | Rosette-forming Diatom | Several | Euplankton; Filter-clogging; Taste & Odour/ Eutrophic |
| Oedogonium sp. | Cyanophyta | Several | Periphyton; stagnant water |
| Mougeotia sp. | Chlorophyta / Zygnematacae | Several | Periphyton/Plankton |
| Spirogyra sp. | Chlorophyta / Zygnematacae | Several | Filter-clogging; Polluted water |
| Pediastrum | Colonial Chlorophyta | Several | Plankton |
| Synedra sp. | Rosette-forming Diatom | Several | Taste & Odour; Eutrophic |
| Gyrosigma sp. | Diatom | Several | Epipelic; stagnant water |
| Meridion sp. | Diatom | Several | Clean water |
| Ankistrodesmus sp. | Diatom | Several | Clean water; |
| Agmenellum sp. | Chlorophyta | Seen once | Polluted water; |
| Melosira sp. | Diatom | Seen once | Filter-clogging; Taste & Odour |
| Surirella sp. | Diatom | Seen once | Clean water; |
| Scenedesmus sp. | Chlorophyta | Seen once | Plankton; Eutrophic |
| Nitzschia sp. | Diatom | Seen once | Polluted water |
| Chlorococcum sp. | Chlorophyta | Seen once | Edaphic |
| Cladophora sp. | Chlorophyta | Seen once | Clean water to eutrophic |
| Cosmarium sp. | Chlorophyta / Desmid | Seen once | Oligotrophic water |
| Staurastrum sp. | Chlorophyta / Desmid | Seen once | Plankton |
| Amoeba | Protista | Seen once | Decaying bottom vegetation |
| Rotifer | Rotifera | Several | Detritus in water |
| Ceratophyllum sp. | Chlorophyta | Seen once | eutrophic |
1Palmer, 1959 and various other sources in References.
Notice that the organisms I found live mostly as attached organisms; some are planktonic or euplanktonic, able to adapt to a periphyton community. Several have been flagged as responsible for taste and odour and filter-clogging. I did not find any toxicity-associated organisms (these belong mostly to the blue-greens). A range of habitat preferences is also shown: from polluted and organically enriched (eutrophic) water to very clean and oligotrophic waters. This is partly because many ubiquitous organisms can live in a wide range of conditions. Organisms shown to prefer clean and oligotrophic waters are particularly important, given they are a limiting factor.
While eutrophic-associated species can and do live in oligotrophic waters, it is less common for clean water organisms to inhabit polluted environments. For this reason, clean water indicators indicate clean water more than polluted water indicators indicate pollution. What this suggests to me—without being definitive—is that the Otonabee River, though showing signs of organic enrichment through its algal blooms in spring and summer, remains fairly oxygenated and only moderately contaminated by urban runoff in the city. With increased toxicity from urban runoff, one would expect to see the clean water species disappear and more pollution-tolerant organisms dominate.

The Diatom Forest of a Regulated River
The Otonabee River is regulated by several hydroelectricity dams, generating stations and locks as it runs its 48.5 km course from Katchewanooka Lake, just north of Lakefield and through the city of Peterborough to Rice Lake.
The name is Ojibwe and translates to “the river that beats like a heart” from ode, which means “heart” and odemgathat which means “boiling water.” Others have translated it to “flashing waters running swiftly.” Both accurately refer to the rapids that animated the northern section of the river from Lakefield to Peterborough before the hydroelectric dams were created in the late 1800s/early 1900s.
According to Kim Reid, curator at the Peterborough Museum and Archives, since the 1800s a number of industries have sprung up along the river to tap its resources: lumber operations, saw and grist mills, distilleries and canoe manufacturing facilities. In the late 1800s, hydroelectric generating stations were built along the river and associated canal and locks.
The river’s flow regime is regulated by several dams. This means that timing, magnitude and frequency of high and low flows no longer match the natural rhythm of the watershed environment. Flow regulation changes the nature of periphyton habitats through speciation and biomass. Low flows increase periphyton biomass, favouring certain species over others, because of increased light, temperature and conductivity. Of course, this is offset through dessication events from drawdown. Periphyton are the dominant micro-community in a riverine ecosystem, providing an important food source and home to a diversity of other life.
Dams dramatically affect rivers and aquatic biota by altering hydrology, sediment transport, nutrient cycling, temperature regimes, and the movement of organisms. One of the main impacts of a dam is the change in flow dynamics: intensity, velocity, direction. By their very nature, dams create more lake-like environments (lentic) from a more river-like one (lotic). This forces aquatic communities to adapt from free-flowing, erosional habitats to depositional environments.
Having a less mobile substrate along with increased temperatures and higher nutrients in river sections behind dams will lead to increased biomass of certain diatoms and the proliferation of filamentous green algae (e.g. Ceratophylum, Spirogyra, Zygnema, and Mougeotia—all currently blooming in the Otonabee River along with the diatom forests. And all responsible for taste and odor of the water.
Taste and Odour of the Diatom Forest
In addition to providing hydroelectric power, the Otonabee River is a source of raw (untreated) drinking water for the City of Peterborough. Particularly in the warm summer months, Peterborough residents notice that their tap water carries a complex taste and odor with hints of mostly earthy mustiness. This is caused by several volatile organic compounds (VOCs) created and released by benthic algae as secondary metabolites associated with growth and reproduction or in response to age, death or environmental stresses. While not harmful, the T&O compounds are detectable even at extremely small concentrations (e.g. parts per trillion).
The most common T&O compounds in summer Otonabee River water include two terpinoids: geosmin and 2-M19 (2-methylisoborneol), secondary metabolites produced by river algae that include the Cyanobacteria, Actinomycetes, Myxophyta, Chlorophyta and diatoms. The most likely T&O culprit in the Otonabee River appears to be three common filamentous Chlorophyta Spirogyra, Zygnema, and Mougeotia—all temporary guests of the river’s diatom forest. During the warm summer months, these filamentous green algae proceed to dislodge from their natal forest to form clouds of green froth and ‘hair’ in the river shallows. As the algal blooms grow and age and die in masses, they release geosmin and 2-M19 into the water.
The diatom forest also provides potentially significant sources of biogenic taste and odor VOCs in a river. They do this in two ways:
- through cell damage and death: catabolic enzymes are activated during cell damage and death from grazing or other environmental stress, producing oxylipins and nor-carotenoids; these act as pheromones, grazer deterrents, teratogens, and toxins.
- Through cell degradation: VOC release from decaying biomass by heterotrophic bacteria, fungi and other microorganisms, producing hydrocarbons, amines, terpines and sulfides.
When diatoms slough off the top-heavy diatom forest and their frustules break apart—particularly during a diatom crash through some disruptive event (e.g. predation, disease, temperature, photolytic stress, dehydration, chemical treatment, or destructive turbulence such as a storm, dam release, etc.)—the diatoms release oxylipins and polyunsaturated fatty acid (PUFA) derivatives, among other things. Oxylipins carry a fishy, rancid, oily or cucumber odor, caused by unsaturated and polyunsaturated aldehydes (PUAs) and other alkenes derived from the fatty acids. They can also cause a ‘grassy-fruity-floral’ odor. The hydrocarbons, amines, terpenes and sulfides released by the degrading diatom mass may smell like solvent, fuel oil or gasoline, acrid burnt fat, and old tobacco.
Diatom species implicated in these odors include Fragilaria, Synedra, Melosira, and Stephanodiscus among others—all identified in the recent scum. Watson and Jüttner noted that fatty acid and PUFA yield at the cell level are influenced by growth phase, carbon source, and environmental factors such as light intensity, temperature, nutrients, heavy metals, chlorine and pesticides. A few studies have shown that some PUAs act as freshwater grazer defence mechanisms; other studies suggest that oxylipins may act as pheromones for some benthic diatoms such as Gomphonema parvulum.

Diatoms use volatile taste and odor compounds to modify cell function (e.g. as antioxidants, pheromones, and autoregulation) and food web interactions (grazer deterrents, inhibitors, toxins against predators, attractants) and in response to stress.
The production and release of volatile taste and odor compounds is natural to river algae and occurs during cell metabolism, growth and eventual death and decay. It is however noteworthy that environmental stresses can escalate and intensify the release of T&O compounds.
This makes sense for the Otonabee River which experiences certain environmental stresses as it runs through agricultural and urban environments. Agriculture activities and urban runoff sources add excessive nutrients to the river and pose additional stresses through contamination by heavy metals, PAHs, PCBs, pesticides, herbicides and other hormone disrupting chemicals. Peterborough Utilities Group acknowledges that as rainwater “flows over lawns and roads into storm sewers and then directly into local waterways [it carries] with it dissolved pesticides, salts, oil, and other untreated waste products. Faulty septic systems adjacent the water course and waterfowl living in the Otonabee River also contribute pollution.” While the treatment process at their water treatment plant effectively treats most of these, the taste and odour linger.
Organic enrichment and nutrient enrichment of the river from fertilizers and urban runoff also create stress by overly increasing the biomass of river algae, leading to blooms and bloom crashes with higher incidence of T&O.
In their 2016 report card of the Otonabee River, Otonabee Conservation gave the river a C-grade (fair) for water quality, based on phosphorus (nutrient) content and benthic invertebrate populations.
References
Diatoms of North America. 2021.
Burfeid Castellanos, Andrea M. 2018. “Ecological Factors and Diatom Diversity at Rivers of the Iberian Mediterranean River Basins: macro-scale, meso-scale and micro-scale. PhD Thesis, University of Barcelona. 233pp.
Munteanu, N. & R. Maly. 1981. “The effect of current on the distribution of diatoms settling on submerged glass slides.Hydrobiologia 78: 273-282.
Otonabee Conservation. 2018. “Otonabee Region Watershed Report Card 2018.” Otonabee Conservation.
Palmer, Marvin C. 1959. “An Illustrated Manual on the Identification, Significance, and Control of Algae in Water Supplies.” U.S. Department of Health, Education, and Welfare, Cincinnati, Ohio. 98pp.
Roemer, Stephen C., Kyle D. Hoagland, and James R. Rosowski. 1984. “Development of a freshwater periphyton community as influenced by diatom mucilages.” Can. J. Bot.62: 1799-1813.
Ross, R., Cox, E.J., Karayeva, N.I., Mann, D.G., Paddock, T.B.B., Simonsen, R. and Sims, P.A. 1979. “An amended terminology for the siliceous components of the diatom cell.Nova Hedwigia, Beihefte 64: 513-533.
Round, F.E., Crawford, R.M. and Mann, D.G. 1990. “The Diatoms. Biology and Morphology of the Genera.” Cambridge University Press, Cambridge. 747pp.
Smolar-Zvanut, Natasa and Matjaz Mikos. “The impact of flow regulation by hydropower dams on the periphyton community in the Soca River, Slovenia. Hydrological Sciences Journal 59(5):1032-1045.
Watson, Susan and Friedrich Jüttner. 2019. “Biological production of taste and odour compounds.” In: “Taste and Odour in Source and Drinking Water: Causes, Controls, and Consequences”, Chapter 3. IWA Publishing 63-112pp.

Nina Munteanu is a Canadian ecologist / limnologist and novelist. She is co-editor of Europa SF and currently teaches writing courses at George Brown College and the University of Toronto. Visit www.ninamunteanu.ca for the latest on her books. Nina’s bilingual “La natura dell’acqua / The Way of Water” was published by Mincione Edizioni in Rome. Her non-fiction book “Water Is…” by Pixl Press(Vancouver) was selected by Margaret Atwood in the New York Times ‘Year in Reading’ and was chosen as the 2017 Summer Read by Water Canada. Her novel “A Diary in the Age of Water” was released by Inanna Publications (Toronto) in June 2020.










3 thoughts on “Smoke on the Water…When The Diatom Forest Turns Into ‘Café Crema’”